Other reported side effects of STN stimulation include depression and weight gain.

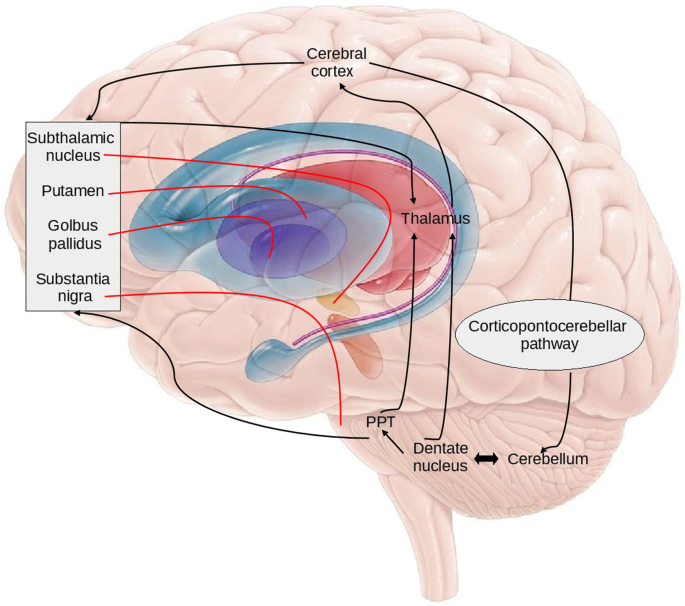
In humans, deep brain stimulation of the STN improves motor function in Parkinson’s disease patients, but has also been shown to cause mild to moderate impairments of performance on cognitive tasks such as verbal fluency, attention, and working memory. Recordings from the rat, cat, and monkey demonstrate that STN neurons encode movements, task-relevant stimuli, and reward presentation. Although initially viewed as an exclusively motor-related structure, converging evidence from both animal and human studies suggests that the STN also regulates cognitive and motivational functions. Deep brain stimulation of the STN (which is thought to functionally inhibit STN output) reduces the motor symptoms of Parkinson’s disease patients, often substantially improving function for as long as five years. Neural activity within the STN increases following the death of the midbrain dopaminergic neurons that causes the movement disorder (for reviews of STN function in human and animal models of Parkinson’s disease, see ). The subthalamic nucleus (STN) is a central node of basal ganglia circuitry that has garnered much attention for its role in Parkinson’s disease.

These data suggest that STN inhibition does not enhance incentive motivation for food in the absence of food restriction and that STN μ-opioid receptors play an important and unique role in motivational processes. In contrast, STN μ-opioid receptor stimulation significantly increased feeding on the palatable diet and reduced the reinforcers earned on a DRL-20 schedule, although DAMGO microinfusions had no effect on PR-2 performance. STN inhibition also did not affect the breakpoint for sucrose pellets during a 1-hr PR-2 reinforcement schedule or impact the number of reinforcers earned on a 1-hr DRL-20 sec reinforcement schedule in non-deprived rats. Although STN inhibition increased ambulatory behavior during 2-hr feeding sessions, it did not significantly alter intake of the sweetened fat diet. Separate behavioral groups (N = 6–9) were tested following bilateral inhibition of the STN with the GABA A receptor agonist muscimol (at 0–5 ng/0.5 μl/side) or following μ-opioid receptor stimulation with the agonist D-Ala 2, N-MePhe 4, Gly-ol-enkephalin (DAMGO at 0, 0.025 or 0.25 μg/0.5 μl/side). In order to further examine the role of the STN on motivation toward food in non-deprived rats, these experiments studied the effects of pharmacological inhibition or μ-opioid receptor stimulation of the STN on the 2-hr intake of a sweetened fat diet, the amount of work exerted to earn sucrose on a progressive ratio 2 (PR-2) schedule of reinforcement, and performance on a differential reinforcement of low-rate responding (DRL) schedule for sucrose reward. The subthalamic nucleus (STN) serves important functions in regulating movement, cognition, and motivation and is connected with cortical and basal ganglia circuits that process reward and reinforcement.
